OVIPOSITION CAGES
1/4 " hardware cloth is used to make a cylindar 12"
h x 8" diameter . Cover top of cylindar with hardware cloth
and also fsaran wrap to confine insects. A hole 1" in diameter is
cut in the top for access to sugar water supply. A 200 ml. pyrex
beaker , filled with 10% sugar water solution , covered with organdy cloth
and secured with a rubberband, is inverted and placed over hole in the
top of the cage. This serves as the food source for the moths. Wax
paper is wrapped around the outside of the cage, and secured with tape.
The moths will oviposition between the hardware cloth and onto the wax
paper. The cage is placed on a tray over a petri dish, containing a layer
of moistened Vermiculite. About 70-95 mixed male and female pupae are placed
on the Vermiculite and allowed to emerge. Keep the vermiculite moist as
this helps adults to emerge with proper wing expansion.
1. Disinfect egg sheet with 10 % clorox solution.
2 .Rinse again with distilled water. Use squirt bottles to gently run
solutions down over eggs.
3. Sheets are then placed between clean towels to dry.
1.Cut up ovipsition sheet into squares which have 12 - 15 eggs / square.
2. Scotch tape( one square to the LID of each diet cup containing Pinto
bean diet. Larvae will hatch and string down onto the diet in the bottom
of the cup, which contains about 3/8 " of media.
3. Place Pinto Beans and water in blender and chop briefly - 2 sec.
Pinto Beans (Ground)
300 gms.
Water, distilled
700 ml.
4. Add next 6 dry ingredients and chop briefly for 1 seconds, 4-5 times
Brewers Yeast ( fortified )
64.0 gms.
Ascorbic Acid
6.0 gms.
Methyl paraben
4.0 gms.
Sorbic Acid
2.0 gms.
Aureomycin, 14.1 % active
1.0 gm.
Propionic Acid, Na salt
1.5 gms.
5. Cook agar and water until thickened, not boiling.
Agar
27.0 gms.
Water, distilled
550.0 mls.
6. Add cooked agar to ingredients in blender and mix thoroughly. Remove
bl ender cover, let media cool 2 minutes before adding vitamin solution.
7. Add vitamin solution and formaldehyde to blender ingredients. Stir
with spoon carefully and then blend thoroughly.
Vitamin Mix
10.0 gms.
Formaldehyde
4.0 mls.
8. Immediately, while hot, transfer media to appropriate containers.
9. Allow media to cool 3 hours or longer until excess moisture has
evaporated from surface before adding larvae and capping.
DIET / PUPATION CUPS
Pupation cups are held in a growth chamber which has a photo period
of 16 : 8 L/D hrs., and a temperature of 78 degrees fahrenheit.
Cups are held for approximately 2 1/2 weeks, at which time
most larvae have pupated and need to be collected. Pull pupae out, webbing
and all.
DISINFECTING PROCEEDURE FOR PUPAE
Place pupae with webbing in a solution of 10% clorox and stir
around gently with your fingers for 60 seconds. This separates the
pupae from the webbing and also disinfects pupae. Rinse pupae carefully
under room temperature tap water, about 60 seconds,until clorox smell is
gone. Place rinsed pupae on several layers of paper towelling. Pull rest
of webbing from pupae. Pupae are now ready to sex and/or use for a new
mating cage.
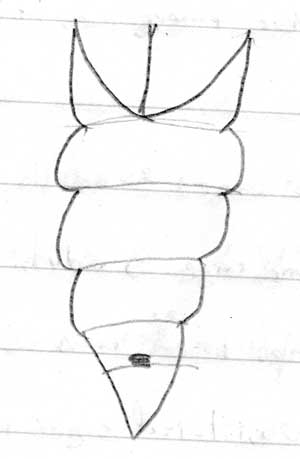
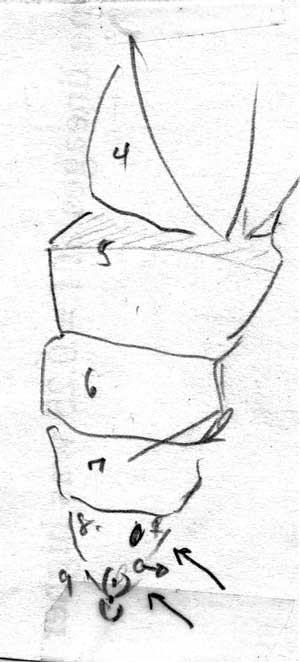 Hold the pupae in good light, count down the segments below the segment covered by the wing buds.
Hold the pupae in good light, count down the segments below the segment covered by the wing buds.
If the forth segment (or 8th segment on A-P axis) is marked, notched, split or therwise not completely intact -> Female; if entire -> Male
Male with a very noticable slit, very close to anal opening, usally on top of perturberance.
--VS 508 Sweetheart Hot Food Cups 1000 / Case
$73.45
--LS8 Sweetheart
Lids
2000 / Case $102.75
Seneca
Bar & Restaurant Supply 20 E. Castle St. Geneva, N. Y. 14456
Phone
# 315-789-2422
--Propionic acid (Sodium salt, 99%) Aldrich Chemical Co., Inc., Milwaukee, WI 53233
--Formaldehyde (certified ACS-F-79, 37% w/w solution)
Fisher Scientific Co., Fair Lawn, NJ 07410
--All other ingredients: Bio-Serv Inc., P.O. Box 450, Frenchtown, NJ
Procedure modified by Andre Levy from protocol by Kathy Poole, NYSAES,
Cornell